|
You probably learned some time ago that chemical bonds are what hold atoms together to form the more complicated aggregates that we know as molecules and extended solids. Before we get into the theory of chemical bonding, we need to define what we are talking about: Exactly what is a chemical bond? And what observable properties can we use to distinguish one kind of bond from another? This is the first of ten lessons that will help familiarize you with the fundamental concepts of this very broad subject. The concept of chemical bonding lies at the very core of Chemistry it is what enables about one hundred elements to form the more than fifty million known chemical substances that make up our physical world. Describe, in a general way, how the infrared spectrum of a substance can reveal details about its molecular structure.State the major factors that determine the distance between two bonded atoms.Explain how the heat released or absorbed in a chemical reaction can be related to the bond energies of the reactants and products.Sketch out a potential energy curve for a typical diatomic molecule, and show how it illustrates the bond energy and bond length. 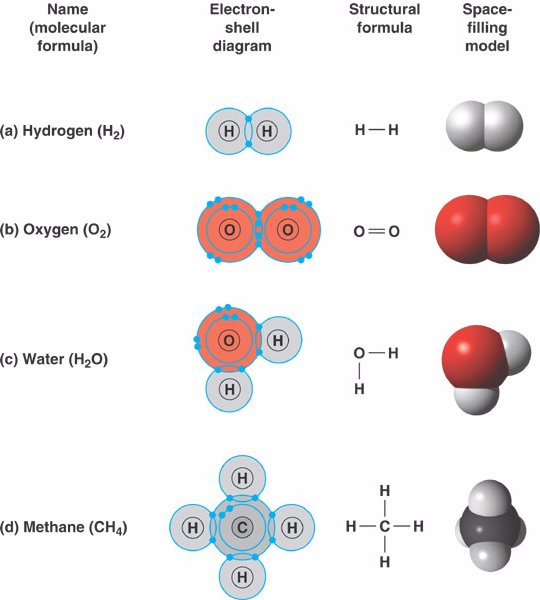  
Explain the difference between stability and reactivity, and how these factors might prevent a given structure from existing long enough to qualify as a molecule.What is meant by the connectivity of a molecule? What additional information might be needed in order to specify its structure?.Make sure you thoroughly understand the following essential ideas which are presented below.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
